 |
Introduction to Ocean Acidification
Ocean acidification is one of the top environmental issues today. It is simply a term that means the oceans are becoming more acidic. This trend is pretty worrying to environmental scientists, as they have studied this to know that almost all ecosystems (particularly those of the upper oceans) will be affected in one way or the other.
 What is ocean acidification? What is ocean acidification?
It is the changes in the chemical make-up of all the waters in the oceans, as well as its temperature, resulting from excess carbon dioxide in the atmosphere. The changes involve the lowering of the pH of the water.
To understand this better, we need to first understand what pH is, together with atmospheric Carbon Dioxide.
 What is pH of water? What is pH of water?
pH of water is the measure of how acidic or basic water is. The scale is from 0 to 14, and 7 is the neutral point (pure water). If the pH is closer to 0, that water is more acidic. If it is closer to 14, the more basic it is.
“During the last 20 years, it has been established that the pH of the world’s oceans is decreasing as a result of anthropogenic carbon dioxide emissions to the atmosphere.” *1
Lower pH seas and oceans are known to cause major problems for marine organisms and ecosystems.
 Carbon Dioxide (CO2) Carbon Dioxide (CO2)
The atmosphere is made up of air, and air is composed of gasses including: Oxygen 21%, Nitrogen 78%, Argon 0.9% and Carbon Dioxide
(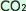 ) 0.04%. These gasses occur naturally in the atmosphere. However, the concentrations of atmospheric ) 0.04%. These gasses occur naturally in the atmosphere. However, the concentrations of atmospheric 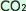 in the atmosphere have increased a lot since the industrial revolution. Humans are burning a lot more fossil fuels than ever before. Fossil fuels are the main source of in the atmosphere have increased a lot since the industrial revolution. Humans are burning a lot more fossil fuels than ever before. Fossil fuels are the main source of 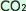 . It is known that the Amazon, together with other major forests covers are large absorbers of . It is known that the Amazon, together with other major forests covers are large absorbers of 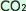 from the atmosphere, but in fact, the Oceans, are the largest carbon sinks on the planet. from the atmosphere, but in fact, the Oceans, are the largest carbon sinks on the planet.
Now we shall see a bit more about how the acidification occurs. Click the next button.

|
 |

![]()
